[Published online Journal of Computer Chemistry, Japan Vol.25, 26-27, by J-STAGE]
<Title:> 動的構造サンプリングに基づくNeuropsinの 反応機構解析:MD×QM/MMアプローチ
<Author(s):> 藤原 麻衣, 阿部 光将, 堀岡 洋太, 福西 快文, 田村 英紀, リントゥルオト ユハ, リントゥルオト 正美
<Corresponding author E-Mill:> masami(at)kpu.ac.jp
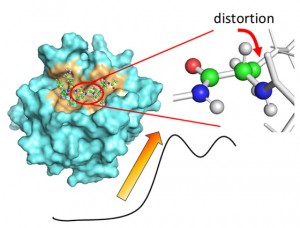
<Abstract:> The enzymatic reaction mechanism of the serine protease neuropsin (KLK8) was investigated using QM/MM calculations combined with MD simulations. The out-of-plane distortion of the scissile amide bond in the substrate-binding complex contributed to a reduction in the activation barrier. Nucleophilic attack by Ser195 Oγ initiated proton transfer among the catalytic triad and the formation of a tetrahedral intermediate. Free energy analysis indicated that this intermediate formation is the rate-determining step, emphasizing the importance of amide bond distortion and oxyanion hole stabilization for catalysis.
<Keywords:> serine protease, neuropsin, KLK8, MD simulation, QM/MM
<URL:> https://www.jstage.jst.go.jp/article/jccj/25/1/25_2025-0028/_article/-char/ja/
<Title:> 動的構造サンプリングに基づくNeuropsinの 反応機構解析:MD×QM/MMアプローチ
<Author(s):> 藤原 麻衣, 阿部 光将, 堀岡 洋太, 福西 快文, 田村 英紀, リントゥルオト ユハ, リントゥルオト 正美
<Corresponding author E-Mill:> masami(at)kpu.ac.jp
<Abstract:> The enzymatic reaction mechanism of the serine protease neuropsin (KLK8) was investigated using QM/MM calculations combined with MD simulations. The out-of-plane distortion of the scissile amide bond in the substrate-binding complex contributed to a reduction in the activation barrier. Nucleophilic attack by Ser195 Oγ initiated proton transfer among the catalytic triad and the formation of a tetrahedral intermediate. Free energy analysis indicated that this intermediate formation is the rate-determining step, emphasizing the importance of amide bond distortion and oxyanion hole stabilization for catalysis.
<Keywords:> serine protease, neuropsin, KLK8, MD simulation, QM/MM
<URL:> https://www.jstage.jst.go.jp/article/jccj/25/1/25_2025-0028/_article/-char/ja/