[Published online Journal of Computer Chemistry, Japan Vol.24, 10-13, by J-STAGE]
<Title:> 塩化亜鉛触媒を用いる芳香族ニトリルへのグリニャール付加反応の理論的研究
<Author(s):> 吉川 武司, 梅澤 美帆, 椿 紗穂里, 坂田 健, 波多野 学
<Corresponding author E-Mill:> takeshi.yoshikawa(at)phar.toho-u.ac.jp
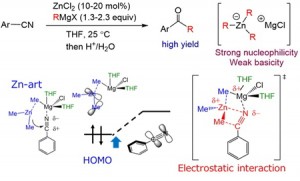
<Abstract:> Alkylation reactions of aromatic nitriles using Grignard reagents produce ketones after hydrolysis. However, this addition reaction is slower than when using reactive organolithium(I) reagents. In the previous paper, we improved the reaction by using zinc(II)ates, which are generated in situ using Grignard reagents and zinc chloride (ZnCl2). The corresponding ketones and amines were obtained in good yields under mild reaction conditions. In this study, the reaction mechanism was theoretically investigated by using density functional theory (DFT). The reactivity with ZnCl2 was verified thorough orbital interaction analysis and non-covalent interactions analysis.
<Keywords:> キーワードGrignard reaction, Schlenk equilibrium, Zn(II)ate complex, Tetrahydrofuran solvent, Density functional theory
<URL:> https://www.jstage.jst.go.jp/article/jccj/24/1/24_2024-0037/_article/-char/ja/
<Title:> 塩化亜鉛触媒を用いる芳香族ニトリルへのグリニャール付加反応の理論的研究
<Author(s):> 吉川 武司, 梅澤 美帆, 椿 紗穂里, 坂田 健, 波多野 学
<Corresponding author E-Mill:> takeshi.yoshikawa(at)phar.toho-u.ac.jp
<Abstract:> Alkylation reactions of aromatic nitriles using Grignard reagents produce ketones after hydrolysis. However, this addition reaction is slower than when using reactive organolithium(I) reagents. In the previous paper, we improved the reaction by using zinc(II)ates, which are generated in situ using Grignard reagents and zinc chloride (ZnCl2). The corresponding ketones and amines were obtained in good yields under mild reaction conditions. In this study, the reaction mechanism was theoretically investigated by using density functional theory (DFT). The reactivity with ZnCl2 was verified thorough orbital interaction analysis and non-covalent interactions analysis.
<Keywords:> キーワードGrignard reaction, Schlenk equilibrium, Zn(II)ate complex, Tetrahydrofuran solvent, Density functional theory
<URL:> https://www.jstage.jst.go.jp/article/jccj/24/1/24_2024-0037/_article/-char/ja/