[Published online Journal of Computer Chemistry, Japan -International Edition Vol.7, -, by J-STAGE]
<Title:> Density Functional Study of σ Bond Cleavage in P P Multiple Bond of Phosphinophosphinidene
<Author(s):> Toshiaki MATSUBARA, Keisuke SHIRASAKA
<Corresponding author E-Mill:> matsubara(at)kanagawa-u.ac.jp
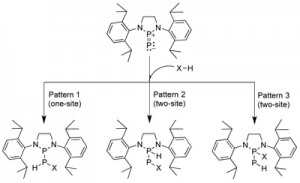
<Abstract:> Recently, the synthesis of phosphinophosphinidene, which is a phosphorus analog of carbene, has been reported. Subsequent experimental reports have shown that phosphinophosphinidene acts as an electron acceptor. Because the terminal phosphorus atom inherently acts as an electron donor, chemical reactions may lead to the σ bond cleavage at the phosphorus atom through charge-transfer interaction. In this study, we explore the possibility of the σ bond cleavage in H H, C H, O H, N H, and B H bonds by means of the density functional method using the model molecules, H2, CH4, H2O, NH3 and BH3. For H2 and CH4, the H H and the C H bonds were found to be broken at the single site of the terminal phosphorus atom by the charge-transfer interactions. The potential energy barrier of about 22 24 kcal/mol is similar to that for carbene, suggesting the possibility of σ bond cleavage in phosphinophosphinidene. In contrast, for H2O and NH3, the O H and N H bonds are broken at the two sites of both phosphorus atoms by the abstraction of hydrogen as a proton. In the case of BH3, cleavage of the B H bond occurs easily at both the single and dual sites of the phosphorus atoms.
<Keywords:> Density functional method, Phosphinophosphinidene, σ Bond cleavage, Reaction mechanism
<URL:> https://www.jstage.jst.go.jp/article/jccjie/7/0/7_2020-0003/_html
<Title:> Density Functional Study of σ Bond Cleavage in P P Multiple Bond of Phosphinophosphinidene
<Author(s):> Toshiaki MATSUBARA, Keisuke SHIRASAKA
<Corresponding author E-Mill:> matsubara(at)kanagawa-u.ac.jp
<Abstract:> Recently, the synthesis of phosphinophosphinidene, which is a phosphorus analog of carbene, has been reported. Subsequent experimental reports have shown that phosphinophosphinidene acts as an electron acceptor. Because the terminal phosphorus atom inherently acts as an electron donor, chemical reactions may lead to the σ bond cleavage at the phosphorus atom through charge-transfer interaction. In this study, we explore the possibility of the σ bond cleavage in H H, C H, O H, N H, and B H bonds by means of the density functional method using the model molecules, H2, CH4, H2O, NH3 and BH3. For H2 and CH4, the H H and the C H bonds were found to be broken at the single site of the terminal phosphorus atom by the charge-transfer interactions. The potential energy barrier of about 22 24 kcal/mol is similar to that for carbene, suggesting the possibility of σ bond cleavage in phosphinophosphinidene. In contrast, for H2O and NH3, the O H and N H bonds are broken at the two sites of both phosphorus atoms by the abstraction of hydrogen as a proton. In the case of BH3, cleavage of the B H bond occurs easily at both the single and dual sites of the phosphorus atoms.
<Keywords:> Density functional method, Phosphinophosphinidene, σ Bond cleavage, Reaction mechanism
<URL:> https://www.jstage.jst.go.jp/article/jccjie/7/0/7_2020-0003/_html